作者简介:刘铁斌,1978年生,男,辽宁省沈阳市人,高级工程师,从事固定床渣油加氢工艺技术研究。
介绍新一代FZC系列渣油加氢处理催化剂的工业应用。新一代FZC系列渣油加氢处理催化剂性能显著提高,催化剂体系具有高的容金属能力和抗结焦能力,活性和稳定性好,能够有效保证装置长周期稳定运行,催化剂整体加氢性能显著提升。工业应用表明,新一代FZC系列渣油加氢处理催化剂能够很好满足用户要求。
The commercial application of new generation FZC residue hydrotreating catalysts by Sinopec Fushun(Dalian) Research Institute of Petroleum and Petrochemicals(FRIPP) was described in this paper.FZC hydrotreating catalysts featured significantly enhanced performance,higher metal-tolerant capacity,higher resistance to coking,and good activity and stability,which secured stable and long-term operation in commercial plants and remarkable increased hytrotreating capability.
渣油的深度加工技术在现代炼油工业中起重要作用, 其中, 渣油固定床加氢处理技术成熟, 作为催化裂化原料加氢预处理工艺, 已在工业上成功应用。渣油加氢与催化裂化联合使用能够实现渣油的清洁高效转化, 适合我国重油加工技术[1, 2, 3]。
中国石油化工股份有限公司抚顺石油化工研究院1986年起开始渣油加氢技术的开发, 1999年S-RHT渣油加氢成套技术首次国产化。经过30年技术的不断积累和进步, 开发出新一代FZC系列催化剂及其配套的工艺技术, 并在国内外10余套渣油加氢处理装置成功应用40多个周期。本文介绍新一代FZC系列渣油加氢处理催化剂的工业应用。
新一代FZC系列渣油加氢处理催化剂牌号和主要功能见表1。
| 表 1 新一代FZC系列固定床渣油加氢处理催化剂 Table 1 New generation FZC series fixed bed residue hydrotreating catalysts |
中国石油化工股份有限公司茂名分公司(茂名石化)2.0 Mt· a-1渣油加氢装置采用S-RHT渣油加氢成套技术设计建造, 反应部分采用双系列同开同停设置, 每系列包括5台反应器。第10周期工业运转Ⅱ 系列采用新一代FZC系列渣油加氢催化剂, Ⅰ 系列装填国外某催化剂。第10周期工业运转于2015年1月16日, 进料量和渣油比基本达到设计要求, 2016年3月17日停工, 运行时间427天。
茂名石化渣油加氢装置第10周期Ⅰ 系列和Ⅱ 系列催化剂平均反应温度的变化如图1所示。
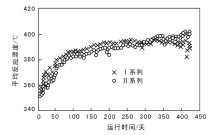 | 图 1 茂名石化渣油加氢装置Ⅰ 系列和Ⅱ 系列催化剂平均反应温度的变化Figure 1 Average reaction temperatures of the first and second series catalysts for residue hydrotreating unit of Maoming Petrochemical Company |
从图1可以看出, 装置运行前300天, Ⅱ 系列平均反应温度较Ⅰ 系列低(4~6) ℃, 装置运行300天后, Ⅰ 系列第一反应器出现热点后无法继续提升温度, 导致平均反应温度低于Ⅱ 系列。
茂名石化渣油加氢装置第10周期Ⅰ 系列和Ⅱ 系列进料量的变化如图2所示。
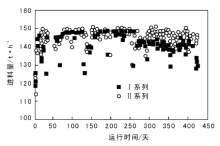 | 图 2 茂名石化渣油加氢装置进料量的变化Figure 2 The change of feed rate in the first and second series of residue hydrotreating unit in Maoming Petrochemical Company |
由图2可见, 运行初期和中期进料量保持一致, 300天后Ⅰ 系列进料量较Ⅱ 系列低(5~10) t· h-1。
茂名石化渣油加氢装置第10周期Ⅰ 系列和Ⅱ 系列加氢产品硫含量、残炭含量和金属(Ni+V)含量的变化如图3所示。
 | 图 3 茂名石化渣油加氢产品硫含量、残炭含量和金属(Ni+V)含量的变化Figure 3 Content tendency of sulfur, carbon residue and metal (Ni+V) in hydrogenation product of Maoming Petrochemical Company |
由图3可以看出, 在装置两个系列进料性质相同条件下, Ⅱ 系列加氢产品平均硫质量分数0.42%, Ⅰ 系列0.48%; Ⅱ 系列加氢产品平均残炭质量分数5.60%, Ⅰ 系列5.87%; Ⅱ 系列和Ⅰ 系列加氢产品金属(Ni+V)含量约15.0 μ g· g-1。Ⅱ 系列在较低反应温度下加氢产品性质更优, 加氢脱硫和残炭加氢转化能力明显优于Ⅰ 系列, 表明新一代FZC系列渣油加氢处理催化剂具有较高的加氢活性和稳定性。
中国石化扬子石油化工有限公司(扬子石化)2.0 Mt· a-1渣油加氢装置采用单系列四台反应器设置方案, 第1周期成功运行450天, 装置第2周期设计值为550天。在装置满足产品指标条件下, 一方面采用新一代FZC系列渣油加氢处理催化剂提升整体容金属能力和高活性催化剂加氢性能; 另一方面对催化剂级配进行优化, 使渣油加氢装置各反应器更加均衡容积金属和积炭。
截至2017年3月17日装置稳定运行480天, 第一反应器入口压力16.9 MPa, 总空速0.20 h-1, 平均反应温度387 ℃, 四台反应器压降趋势平稳, 平均反应温度和各反应器压降趋势分别如图4~5所示。
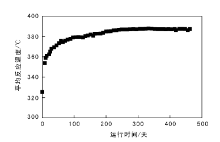 | 图 4 扬子石化渣油加氢装置平均反应温度的变化Figure 4 The tendency of average reaction temperatures of residue hydrotreating unit in Yangzi Petrochemical Company |
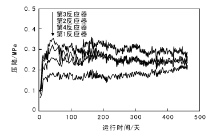 | 图 5 扬子石化渣油加氢装置反应器压降的变化Figure 5 The tendency of the pressure drop of each reactor of residue hydrotreating unit in Yangzi Petrochemical Company |
扬子石化渣油加氢原料油与加氢常渣硫含量、残炭含量及金属(Ni+V)含量的变化如图6所示。
 | 图 6 扬子石化渣油加氢原料油与加氢常渣硫含量、残炭含量及金属(Ni+V)含量的变化Figure 6 Content tendency of sulfur, carbon residue and metal (Ni+V) of feed and product in residue hydrotreating unit of Yangzi Petrochemical Company |
由图6可以看出, 扬子石化渣油加氢处理装置原料平均硫质量分数3.20%, 加氢常渣平均硫质量分数0.42%(设计值0.45%)。原料平均残炭质量分数11.97%, 加氢常渣平均残炭质量分数5.32%(设计值5.7%)。原料平均金属(Ni+V)含量78.96 μ g· g-1, 加氢常渣平均金属(Ni+V)含量14.45 μ g· g-1(设计值15.00 μ g· g-1)。由此可以看出, 加氢常渣各项杂质指标能够满足设计指标要求, 是优质的催化裂化进料。
(1) 新一代FZC系列渣油加氢催化剂表现出良好的加氢活性和稳定性, 加氢常渣是优质的催化裂化进料, 实现了重油的清洁高效转化。
(2) 新一代FZC系列渣油加氢催化剂具有高的容金属能力和抗结焦能力, 能够有效保证装置长周期稳定运行。
The authors have declared that no competing interests exist.
| [1] |
|
| [2] |
|
| [3] |
|

